|
Back to Blog
Antimony number of valence electrons5/3/2023 The electronic configuration of Selenium will be 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p4. How do you write the electron configuration for Selenium? What is the electronic configuration of Selenium 34? What is the boiling Point of Selenium in Kelvin?īoiling Point of Selenium in Kelvin is 958 K. Melting Point of Selenium in Kelvin is 494 K. What is the melting Point of Selenium in Kelvin? Nitrogen, phosphorus, and arsenic can form ionic compounds by. What is the boiling Point of Selenium?īoiling Point of Selenium is 958 K. The Group 5A elements have five valence electrons in their highest-energy orbitals (ns2np3). 
Selenium has 34 electrons out of which 6 valence electrons are present in the 4s2 4p4 outer orbitals of atom. How many valence electrons does a Selenium atom have? The element Selenium was discovered by J. It is located in group 16 and period 4 in the modern periodic table. Selenium is the 34 element on the periodic table. Selenium is a chemical element with the symbol Se and atomic number 34. What is the position of Selenium in the Periodic Table? Selenium is a chemical element with symbol Se and atomic number 34. To form abbreviated notation of electronic configuration, the completely filled subshells are replaced by the noble gas of the preceding period in square brackets. The abbreviated electronic configuration of Selenium is 3d10 4s2 4p4. Furthermore, all pnictogens have five valence electrons two of these electrons are paired and exist in the s subshell, while the remaining three electrons exist in the p shell, unpaired. What is the abbreviated electronic configuration of Selenium? Antimony can react with almost all of the metals on the periodic table to form pnictides. The electronic configuration of Selenium is 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p4. What is the electronic configuration of Selenium? Selenium Thermal Properties - Enthalpies and thermodynamics  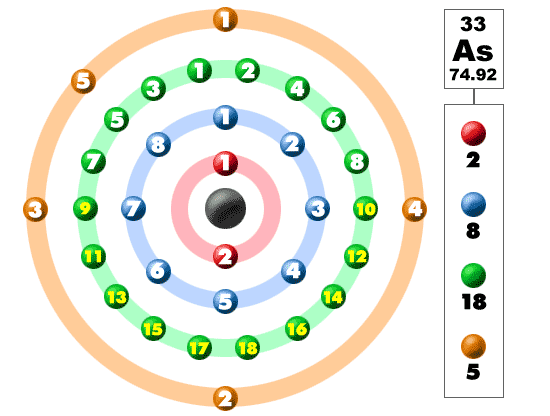
Optical Properties of Selenium Refractive IndexĪcoustic Properties of Selenium Speed of Sound Selenium Magnetic Properties Magnetic Type Selenium Heat and Conduction Properties Thermal Conductivity Refer to table below for the Electrical properties ofSelenium Electrical Conductivity Hardness of Selenium - Tests to Measure of Hardness of Element Mohs Hardness Phenom., 1980, 21, 275.Refer to below table for Selenium Physical Properties DensityĤ.819 g/cm3(when liquid at m.p density is $3.99 g/cm3) Mårtensson, "Core-Level Binding Energies in Metals," J. Lide, (Ed.) in Chemical Rubber Company handbook of chemistry and physics, CRC Press, Boca Raton, Florida, USA, 81st edition, 2000. China produces 88 of the world’s antimony. Antimony can also be found as the native metal. It is extracted by roasting the antimony(III) sulfide to the oxide, and then reducing with carbon. It is most often found as antimony(III) sulfide. Ley, Eds., Photoemission in Solids I: General Principles (Springer-Verlag, Berlin) with additional corrections, 1978. Antimony is not an abundant element but is found in small quantities in over 100 mineral species. 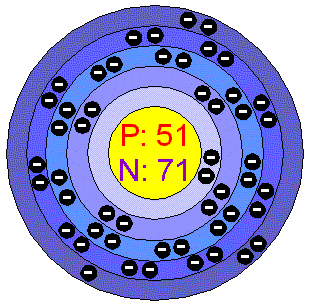
Burr, "Reevaluation of X-Ray Atomic Energy Levels," Rev. They are tabulated elsewhere on the WWW (reference 4) and in paper form (reference 5). The data are adapted from references 1-3. I am grateful to Gwyn Williams (Jefferson Laboratory, Virginia, USA) who provided the electron binding energy data. The binding energies are quoted relative to the vacuum level for rare gases and H 2, N 2, O 2, F 2, and Cl 2 molecules relative to the Fermi level for metals and relative to the top of the valence band for semiconductors. All values of electron binding energies are given in eV. 1967, 47, 1300.Įlectron binding energies Electron binding energies for antimony. These effective nuclear charges, Z eff, are adapted from the following references: Antimony exists in many allotropic forms (physically distinct conditions that result from different arrangements of the same atoms in molecules or crystals ). Effective nuclear charges for antimony 1s antimony (Sb), a metallic element belonging to the nitrogen group (Group 15 Va of the periodic table ).
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
